
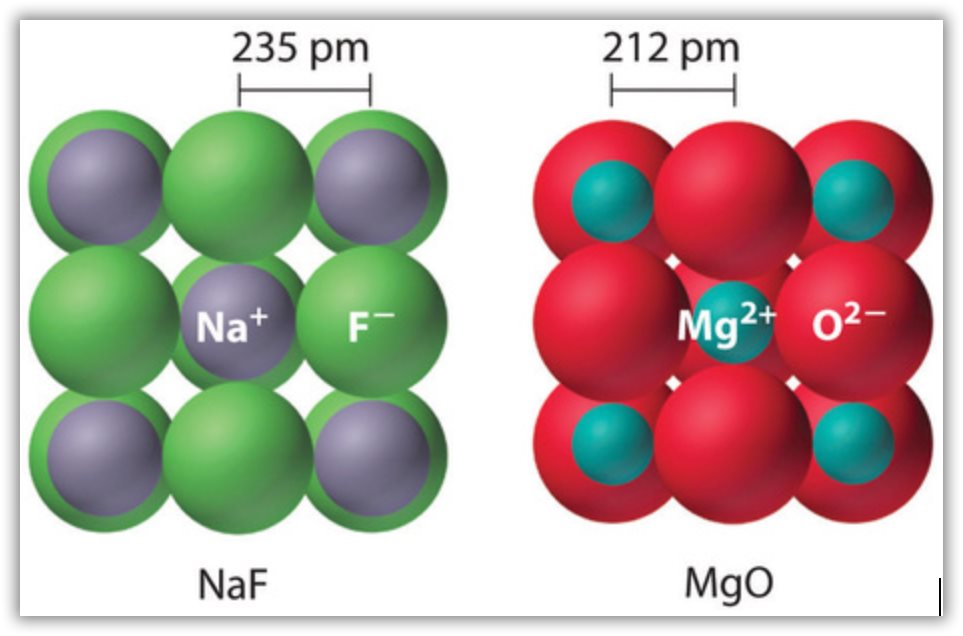
Why does lattice energy increase down a group?Īs we move down the group, the lattice energy decreases. Hence, the lattice energy of MgO is 3790 kJ. The various steps involved in this process are stated below. How do you find the lattice energy of MgO?Īnswer and Explanation: The lattice energy of MgO is given by equation MgO(s)→Mg2+(g)+O2−(g) M g O ( s ) → M g ( g ) 2 + + O ( g ) 2 −. How do you find the lattice energy using the Born Haber cycle? Which has more lattice energy NaCl or Mgcl2?Īlcl3>Mgcl2>Nacl because lattice energy directly proportional to charge on the cation. Due to this, the attraction between them decreases and finally the less lattice energy released during the process. This is because with the when the size of ions increase, the distance between their nuclei increases. When the radius of ions increases, the lattice energy decrease. How does lattice energy depends on ion size? Lattice energy increases for ions with higher charges and shorter distances between ions. Mg2+ ion is smallest in size and thus has highest lattice energy.įor ionic bonds, the lattice energy is the energy required to separate one mole of a compound into its gas phase ions.
0 Comments
Leave a Reply. |



 RSS Feed
RSS Feed
